In natural aquatic systems and contaminated environments, the environmental fate of many toxic metals and metalloids is not governed solely by well-crystallized minerals. Instead, highly reactive and structurally disordered amorphous phases often act as critical intermediates that regulate contaminant transformation and mobility. Despite their importance, the formation mechanisms and kinetic pathways of these amorphous materials remain poorly understood due to the difficulty of characterizing their transient structures and dynamic evolution.
To address this challenge, a research team from the School of Earth System Science at Tianjin University investigated amorphous ferric arsenate (AFA), a key mineral phase controlling arsenic sequestration in environmental systems. Using in situ synchrotron-based small-angle X-ray scattering (SAXS), the team directly monitored the precipitation process of AFA and identified a previously unrecognized two-stage nonclassical formation pathway, fundamentally different from classical nucleation theory.
A nonclassical pathway for amorphous mineral formation
The results show that AFA formation does not proceed through the direct addition of ions or molecules to a growing nucleus. Instead, the process involves a hierarchical particle-assembly mechanism.
Initially, nanoscale precursor particles form in solution and aggregate into loosely organized clusters. Subsequent growth occurs primarily through particle attachment, leading to the development of an amorphous structure characterized by mass-fractal organization.
This mechanism provides a potential general framework for understanding the formation of widely occurring environmental amorphous phases, including iron, manganese, and aluminum hydroxides. The findings highlight the importance of particle-mediated aggregation processes in controlling amorphous mineral formation in natural geochemical systems.
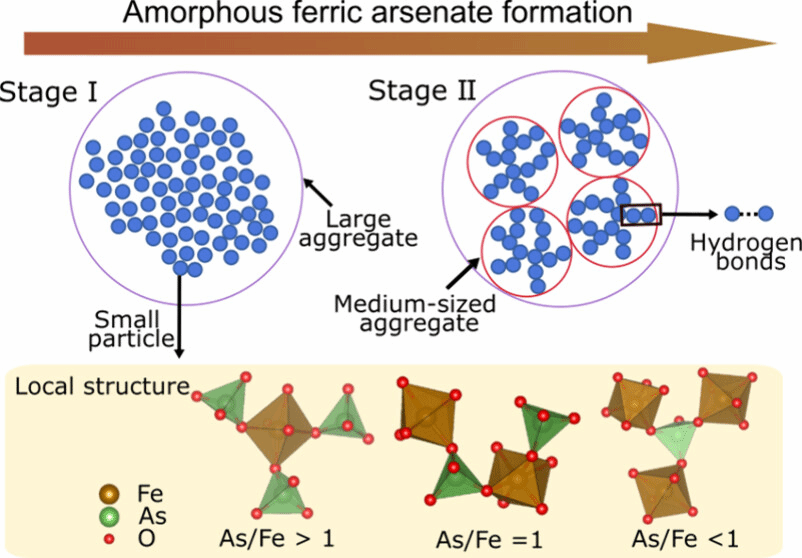
Figure 1. Schematic diagram of the two-stage formation pathway of amorphous ferric arsenate (AFA).
Environmental controls on arsenic sequestration
The study further demonstrates that key environmental variables—including pH, temperature, and the arsenic-to-iron ratio—regulate AFA precipitation kinetics by influencing the stability and interactions of precursor particles.
These results explain why arsenic exhibits markedly different mobility and immobilization behaviors across environmental settings. By linking microscopic formation mechanisms with macroscopic environmental observations, the work improves predictive understanding of arsenic biogeochemical cycling in natural waters and soils.
Implications for arsenic remediation strategies
Beyond fundamental geochemical insights, the findings also have implications for environmental remediation. The study suggests that regulating solution chemistry—such as optimizing the arsenic-to-iron ratio and pH—can steer amorphous mineral formation toward pathways that maximize arsenic immobilization.
Based on this concept, the authors propose a new strategy for arsenic pollution control: promoting rapid formation of amorphous ferric arsenate to initially immobilize large fractions of dissolved arsenic, followed by subsequent purification or stabilization processes. This approach provides a process-based theoretical framework for developing low-energy and high-efficiency in situ arsenic stabilization technologies.